Quality/Supplier Quality
Supplier Quality Engineer
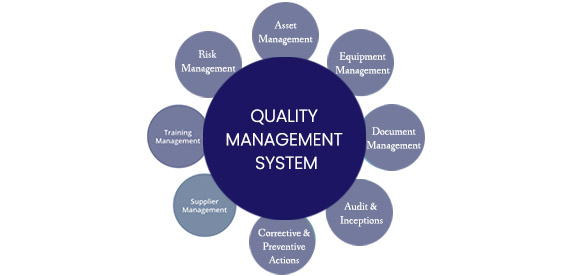
Implementing an effective QMS has always been considered an essential requirement for a manufacturer to maintain and improve its product and service quality. Globally many regulatory authorities incorporate Quality Management System as one of the mandatory requirements for pharmaceutical drugs/products and medical devices. QMS will not only help companies but develop a culture of quality, support data integrity, perform risk calculations, keep suppliers under control, and maintain overall compliance.
To meet this requirement, you can count on Career Prime resources, who can support on but not limited to:
- QMS compliances to 21 CFR 210/211, 21 CFR 820, ISO 9001, and ISO 13485
- Gap Analysis and DHF Remediation
- Test Method Validation, Change Control Management
- Quality Audits, Risk Management System (ISO14971), FMEA
- Supplier Quality, Sampling Plans, Documentation and Design Controls
- 21 CFR 820, ISO 13485 regulations, ISO 9001
- NCR’S, CAPA, Product and Process Controls
- SOP’s, Work Instructions, Control Plans
“Your trust – Our Quality”
At Career Prime, we understand the needs of clients, thus we strive to supply the best of the consultants to clients as per their skillsets and experience.
